 |
Reconstitution of the immune system after treatment for cancer has been studied extensively, particularly after HSCT.3-5 Immunocompetence returns more rapidly in patients undergoing nontransplant remission-induction chemotherapy than in patients who receive an HSCT, and defines the period the patient is at risk of infection. Risk of IFI in nontransplant patients is usually limited to the period of neutropenia, typically 3 to 4 weeks, whereas in HSCT patients, the risk period extends beyond the period of neutropenia and persists for as long as the immune system is compromised. Transplant recipients typically exhibit a predictable pattern of immune system deficiency and recovery (Fig 2).

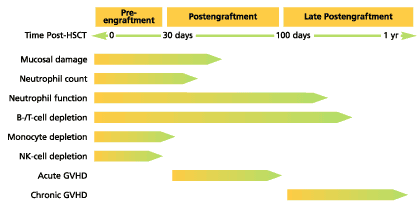
Patients present for transplantation with varying degrees of immune system dysfunction related to previous treatment for underlying disease. Neutropenia associated with anticancer drugs such as cyclophosphamide, fluorouracil, and vinblastine may be present. Other drugs frequently given, including prednisone, inhibit the humoral and cell-mediated immune responses. Irradiation and malnutrition decrease T-lymphocyte function.
The first 30 days after transplantation, the preengraftment phase, begins with the myeloablative conditioning regimen initiated immediately before stem-cell infusion. Normal hematopoiesis for neutrophils, monocytes, and macrophages is destroyed. Profound granulocytopenia occurs and virtually all patients lose T and B lymphocytes; with this, the immune memory accumulated through lifetime exposure to infectious agents is also lost. Engraftment is defined as the point at which a patient can maintain a sustained absolute neutrophil count (ANC) of >500/mm3 and a platelet count of >20,000/mm3 for 3 consecutive days without transfusions. A study facilitated by the National Marrow Donor Program of 5,246 patients who underwent unrelated-donor HSCT between August 1991 and June 1999 reported that myeloid engraftment occurred a median of 18 days after marrow infusion.6 Overall, 84% of patients achieved an ANC >500/mm3 by day 28.
Neutrophil bactericidal and oxidative functions lag behind the increase in cell numbers.7 Chemotaxis can be impaired for up to 4 months.8 Corticosteroid use can impair the restoration of phagocytic function. Monocyte and natural killer (NK) cell numbers and function quickly become normal,5 whereas T- and B-lymphocyte counts remain low. Donor lymphocytes from the stem-cell infusion convey passive T-cell immunity during this period, as may lymphocytes from the host that were able to survive the cytotoxic conditioning.
A major defect in host defenses occurring as a side effect of the conditioning regimen is damage to mucosal progenitor cells, causing a loss of epithelial integrity of the gastrointestinal tract, oropharynx, and respiratory tract. Mucositis occurs in 70% of patients undergoing allogeneic transplant.9 Resolution usually begins by day 20 unless complicated by the development of acute GVHD. Patients who receive non-myeloablative or reduced-toxicity conditioning regimens typically have a shorter period of neutropenia and reduced severity of mucositis.
In the period from day 30 to 100 after HSCT, the postengraftment phase, profound neutropenia generally abates and mucosal barriers begin to repair themselves. However, delayed engraftment may prolong neutropenia, and neutrophil and macrophage function will still be suboptimal. A major period of immune deficiency occurs between days 50 and 100, during which time native and passively transferred immunity wanes and is replaced by donor-related immune function. The presence of GVHD will delay immune recovery and perpetuate mucosal abnormalities.
Throughout the late postengraftment period (day 100 to 1 year after transplant), the host defects caused by myelosuppressive conditioning continue to resolve, although it normally takes up to 12 months for normal immune function to return. At this time chronic GVHD may occur, significantly altering anatomical barriers and causing severe mucositis, in association with cellular and humoral immunodeficiences.
Course Number: V035D
This CME Expires on July 1, 2005; no tests will be accepted after this date.
This course is accredited by
The University of Pittsburgh School of Medicine, Center for Continuing Education and The International Immunocompromised Host Society
|
 |

